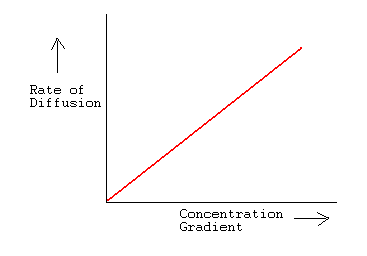
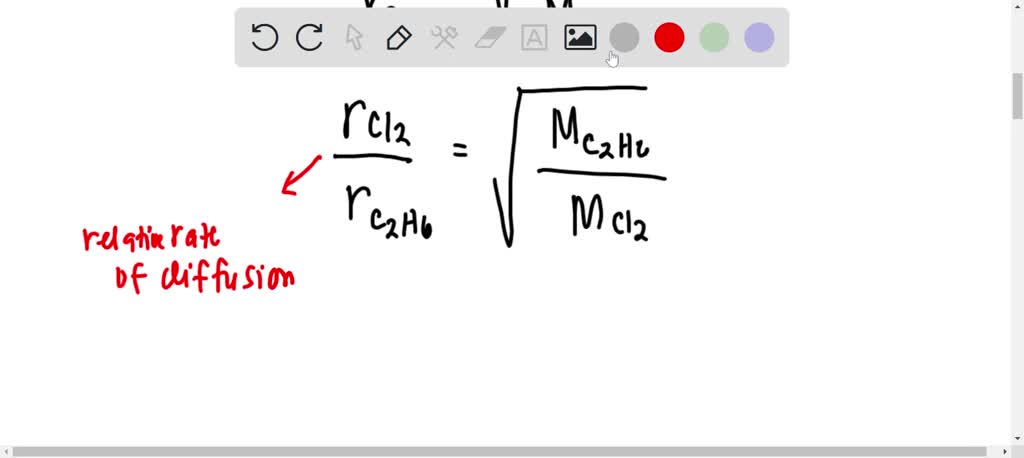
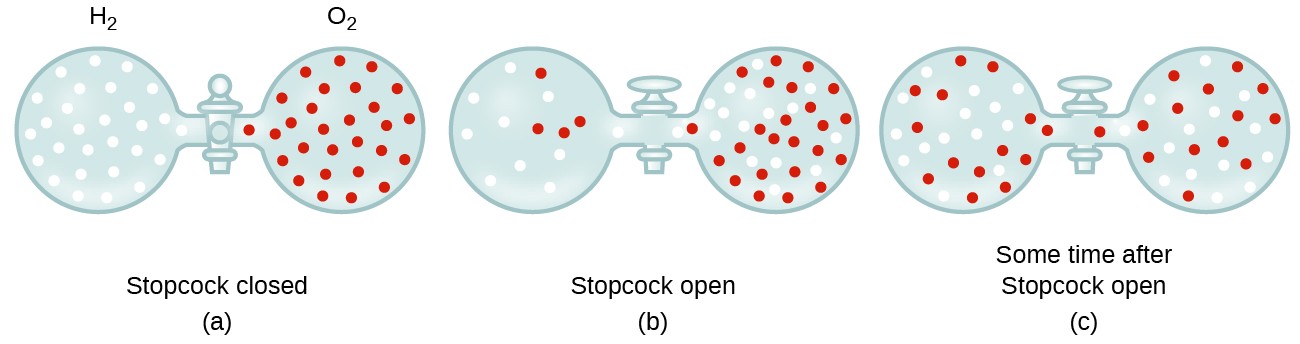
acid base - What is the relative rate of diffusion of ammonia to hydrogen chloride, both in gaseous states? - Chemistry Stack Exchange

The ratio of rate of diffusion of gases A and B is 1 : 4. If the ratio of their masses present in the mixture is 2 : 3, what is the ratio of their mole fraction ?

The ratio of rate of diffusion of gases A and B is `1 : 4`. If the ratio of their masses present in - YouTube

The rate of diffusion of a gas X is √(2) times that of Y . If the molecular weight of X is 16 , the molecular weight of Y is:

If the ratio of the rates of diffusion of two gases A and B is 4:1 the ratio of their density is - YouTube